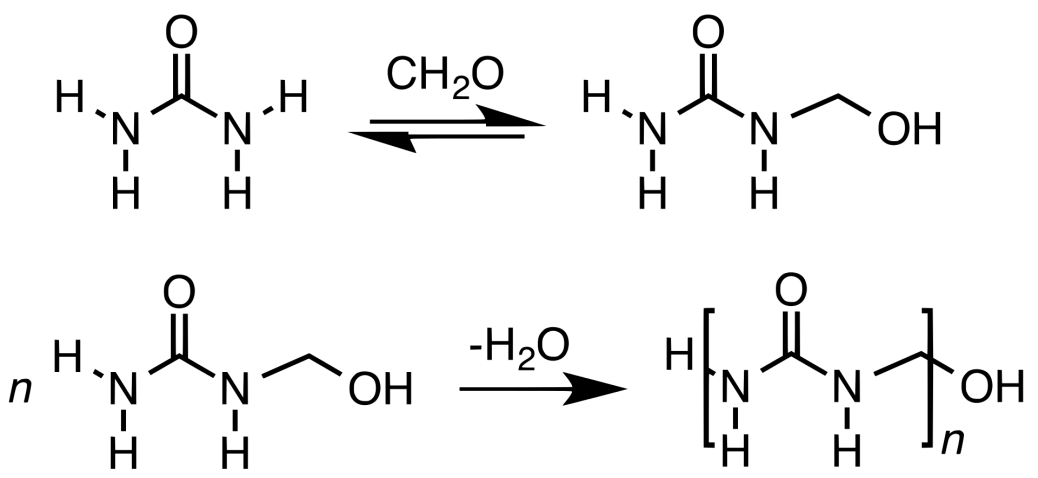
Urea-formaldehyde is pervasive. Examples include decorative laminates, textiles, paper, foundry sand molds, wrinkle resistant fabrics, cotton blends, rayon, corduroy, etc. It is also used to glue wood together. Urea formaldehyde was commonly used when producing electrical appliances casing (e.g. desk lamps). Foams have been used as artificial snow in movies.
Agricultural use
Urea formaldehyde is also used in agriculture as a controlled release source of nitrogen fertilizer. Urea formaldehyde’s rate of decomposition into CO2 and NH3 is determined by the action of microbes found naturally in most soils. The activity of these microbes, and, therefore, the rate of nitrogen release, is temperature dependent. The optimum temperature for microbe activity is approximately 70-90 °F (approx 20-30 °C).
Foam insulation
Urea-formaldehyde foam insulation (UFFI) dates to the 1930s and made a synthetic insulation with R-values near 5.0 per inch. It is a foam, like shaving cream, that is easily injected or pumped into walls. It is made by using a pump set and hose with a mixing gun to mix the foaming agent, resin and compressed air. The fully expanded foam is pumped into areas in need of insulation. It becomes firm within minutes but cures within a week. UFFI is generally found in homes built before the 1970s, often in basements, crawl spaces, attics, and unfinished attics. Visually it looks like oozing liquid that has been hardened. Over time, it tends to vary in shades of butterscotch but new UFFI is a light yellow color. Early forms of UFFI tended to shrink significantly. Modern UF insulation with updated catalysts and foaming technology have reduced shrinkage to minimal levels (between 2-4%). The foam dries with a dull matte color with no shine. When cured, it often has a dry and crumbly texture.




![91{(2%0WF8MDR[$]P`AZV_D.png](https://lin-chemical.com/wp-content/uploads/2017/05/9120wf8mdrpazv_d.png?w=273&h=407)
![P}$H7@[K@624USUOR]ASYYQ.png](https://linchemical4ag.files.wordpress.com/2017/05/ph7k624usuorasyyq.png?w=279&h=423)